Dr. Latha Diwakar

Biosketch
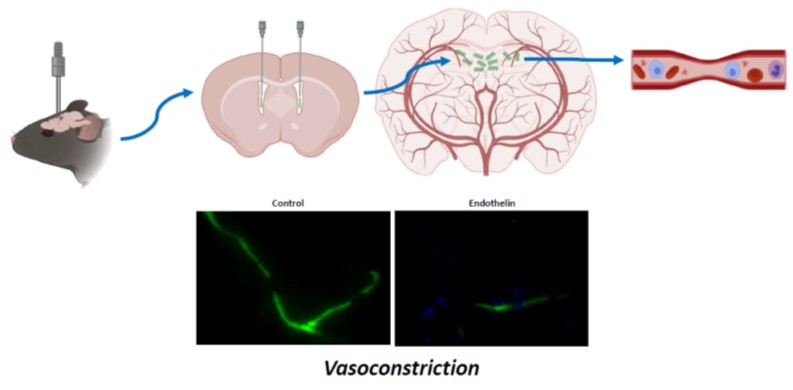
Verma A, Ray A, Bapat D, Diwakar L, Kommaddi RP, Schneider BL, Hirsch EC, and Ravindranath V. (2020), Glutaredoxin 1 Downregulation in the Substantia Nigra Leads to Dopaminergic Degeneration in Mice. Movement Disorders. doi:10.1002/(ISSN)1531-8257. 2020.
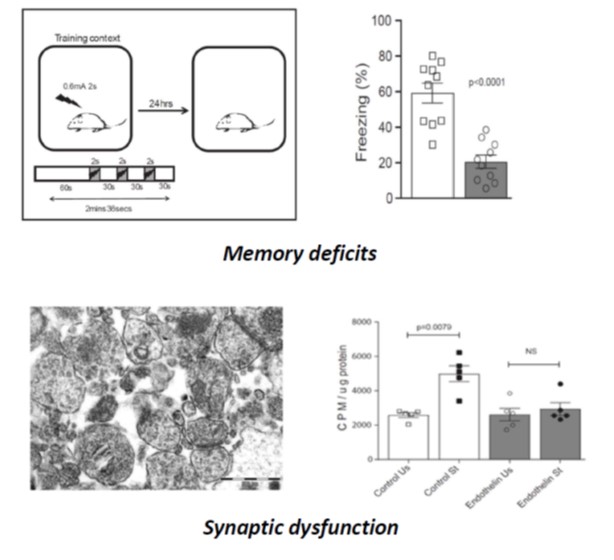
Ahmad F, Das D, Kommaddi RP, Diwakar L, Gowaikar R, Rupanagudi KV, Bennett DA, Ravindranath V. Isoform-specific hyperactivation of calpain-2 occurs presymptomatically at the synapse in Alzheimer’s disease mice and correlates with memory deficits in human subjects. Sci Rep. 2018 Sep 3; 8(1):13119.
S Shenvi, KR Kiran, K Kumar, Diwakar L, GC Reddy. Synthesis and biological evaluation of boswellic acid-NSAID hybrid molecules as anti-inflammatory and anti-arthritic agents. European journal of medicinal chemistry 98, 170-178. 2015.
Samaga L, KK, Rao GV, Reddy GC, Kush AK, Diwakar L. Synthetic racemates of abyssinone I and II induces apoptosis through mitochondrial pathway in human cervix carcinoma cells. Bioorganic Chemistry 56: 54-61. 2014.
Shenvi S, Kumar K, Hatti KS, Rijesh K, Diwakar L, Reddy G.C. Synthesis, anticancer and antioxidant activities of 2,4,5-trimethoxy chalcones and analogues from asaronaldehyde: Sturcture-activity relationship. European Journal of Medicinal Chemistry 62 435-442. 2013.
Diwakar L, Rudresh Kumar KJ, Bachnalkar A, Ravindranath V, Christopher R, Nagaraja D. The influence of MTR A2756G polymorphism on plasma homocysteine in young south Indians. ClinChimActa. 395, 172-174. 2008.
Diwakar L, Ravindranath V. Inhibition of cystathionine-gamma-lyase leads to loss of glutathione and aggravation of mitochondrial dysfunction mediated by excitatory amino acid in the CNS. Neurochem Int. 50, 418-426. 2007.
Diwakar L, Kenchappa RS, Annepu J, Saeed U, Sujanitha R, Ravindranath V, Down-regulation of glutaredoxin by estrogen receptor antagonist renders female mice susceptible to excitatory amino acid mediated complex I inhibition in CNS. Brain Res.1125(1):176-184. 2006.
Diwakar L, Kenchappa RS, Annepu J, Saeed U, Sujanitha R, Ravindranath V, Down-regulation of glutaredoxin by estrogen receptor antagonist renders female mice susceptible to excitatory amino acid mediated complex I inhibition in CNS. Brain Res.1125(1):176-184. 2006.
Kenchappa RS, Diwakar L, Annepu J, Ravindranath V. Estrogen and neuroprotection: higher constitutive expression of glutaredoxin in female mice offers protection against MPTP-mediated neurodegeneration. FASEB J. 18:1102-1114. (Co-first author). 2004.
Devi L, Diwakar L, Raju TR, Kutty BM. Selective neurodegeneration of hippocampus and entorhinal cortex correlates with spatial learning impairments in rats with bilateral ibotenate lesions of ventral subiculum. Brain Res. 960:9-15. 2003.
Kenchappa RS, Diwakar L, Boyd MR, Ravindranath V. Thioltransferase (glutaredoxin) mediates recovery of motor neurons from excitotoxic mitochondrial injury. J Neurosci. 22:8402-8410. 2002.
-
Centre for Brain Research
Indian Institute of Science Campus
CV Raman Avenue
Bangalore 560012, India. - +91 80 2293 3588


